Sybodies neutralise SARS-CoV-2 variants
Synthetic antibodies neutralise different virus variants and prevent them developing resistance.
Since it broke out in 2019, the SARS-CoV-2 pandemic has been dominated by a number of variants of concern (VOCs). The medicines and vaccines used to date have become less effective in the face of viral mutations, while modified treatments have simply caused fresh mutations. The team of researchers led by Philippe Plattet and Markus Seeger at the University of Bern and University of Zurich describe one way of dealing with this war of attrition in their recent publication. They have developed a pair of special antibodies that recognise two different epitopes (part of the surface of an antigen) on the virus and could thus be instrumental in reducing the development of resistant variants.
The spike protein of the Covid-19 virus is one such antigen. It mediates viral entry by binding to the ACE2 receptor on the surface of human cells. The spike protein is also frequently the target of vaccines and medications to treat the disease. Observations show that it can mutate very quickly and varies greatly between variants. The consequences range from loss of treatment efficacy to resistant mutants.
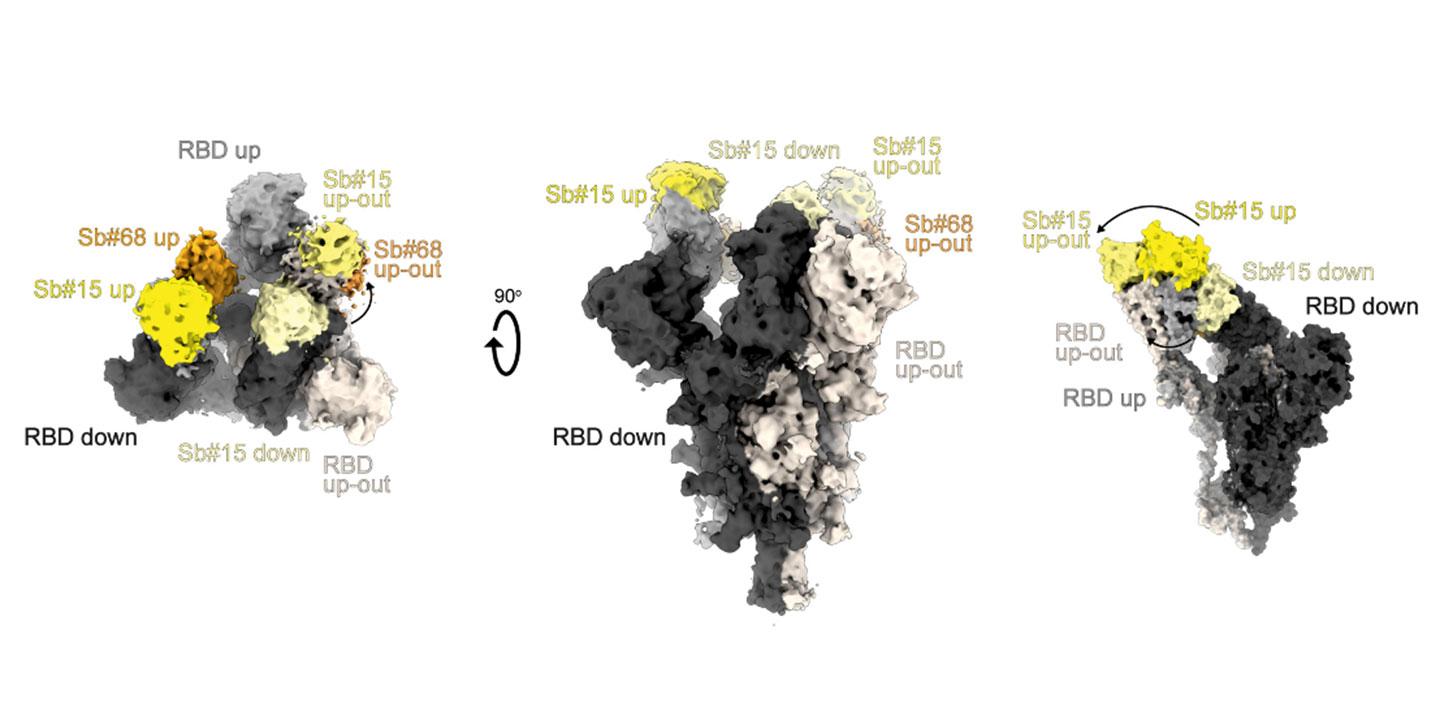
The researchers have now developed two sybodies – Sb#15 and Sb#68 – capable of binding the spike protein to different epitopes at the same time. Sybodies are synthetic nanobodies, in other words antibodies that have just one domain and are very small and stable as a result. Experiments have shown that Sb#15 and Sb#68 compete successfully with ACE2 for the spike protein’s binding sites. Furthermore, the two sybodies were capable of neutralising virus infection in vitro. To enhance their potency, the two sybodies were joined using a linker sequence. GS4, the resulting fusion protein, demonstrated not only greater binding affinity for the viral spike protein, but also a neutralisation rate 100 times greater than that of a single sybody.
It is interesting to note that viral escape mutants rapidly emerged when grown in presence of Sb#15 or Sb#68, whereas this was not observed with GS4. In a further step, a third binding domain was added to GS4, resulting in a construct named Tripod-GS4r. This resulted in a 1000-fold increase in overall neutralisation activity against SARS-CoV-2 VOCs compared with a single sybody.
The results of the study suggest a novel treatment strategy capable of restricting the evolution of new variants in future. Sybodies are not only relatively straightforward and quick to develop, they are also inexpensive. Since they are also simple to use, they are suitable for self-administered inhalable formulations that directly target nasal and lung tissue. Furthermore, the study corroborates the highly promising strategy of binding several viral epitopes at the same time. Marketed antibody cocktails have recently shown that it is possible to slow down the process by which new VOCs evolve and to effectively inhibit viral escape.
Publication
